ISS: Foam-Coarsening Experiment
Science
ISS Utilization: Foam-Coarsening Experiment
March 9, 2020: The Sample Cell Unit S/N 6 of the Foam Coarsening experiment is used for commissioning on the International Space Station. It contains three sample cells filled with liquid. By shaking the pistons inside the cell (the white and black items at the bottom of the cells are the most visible parts of the pistons), foam is generated. The white cell (in third position) contains a humidity sensor. 1)

- The Foam-Coarsening experiment, developed by Airbus for ESA, is scheduled to be activated this month in the Fluid Science Laboratory in the European Columbus module.
- The foams come in self-contained cells and hold liquids that will be shaken (not stirred) and analyzed with laser optics and high-resolution cameras. Researchers are keen to observe how foams behave in microgravity.
- On Earth, the mixture of gas and liquid that makes up a foam quickly starts to change. Gravity pulls the liquid between the bubbles downwards, and the small bubbles shrink while the larger ones tend to grow at the expense of others. Due to drainage, coarsening (or enlarging) and rupture of the bubbles, a foam starts to collapse back to a liquid state.
- But in space foams are more stable because there is no drainage in weightlessness. This allows scientists to study the slower phenomena of a bubble becoming bigger and bursting, which on Earth are masked by the drainage that destabilizes the foam.
- ESA astronaut Frank De Winne performed the Foam-Stability experiment in 2009 by shaking liquid solutions and recording what happened next. The samples ranged from pure water to protein-based fluids, like the ones used for chocolate foams, and antifoaming agents.
- After just ten seconds, the fluids stabilized more quickly and produced more foam than on Earth. Scientists discovered that it was possible to create super-stable foams in zero gravity.
- Building on this extensive foam research, Foam-Coarsening will investigate foam behavior at different liquid stages, particularly as it transitions from a solid- to liquid-like state.
- The results from this research will not just apply to the foam in your morning cappuccino. Foams are used in a wide range of areas from food production to cleaning and sealing products, cosmetics and personal hygiene products, and even construction.
Some Background
At the end of 2009 and in early 2010 a sealed cell, for foam generation and observation, has been designed and manufactured at Astrium Friedrichshafen facilities. With the use of this cell, different sample compositions of "wet foams" have been optimized for mixtures of chemicals such as water, dodecanol, pluronic, aethoxisclerol, glycerol, CTAB (Cetyl Trimethyl Ammonium Bromide), SDS (Sodium Dodecylsulfate), as well as glass beads. This development is performed in the frame of the breadboarding development activities of the Experiment Container FOAM-C for operation in the ISS Fluid Science Laboratory (ISS). The sample cell supports multiple observation methods such as: Diffusing-Wave and Diffuse Transmission Spectrometry, Time Resolved Correlation Spectroscopy and microscope observation, all of these methods are applied in the cell with a relatively small experiment volume <3cm3. These units, will be on orbit replaceable sets, that will allow multiple sample compositions processing (in the range of >40). 2)
Foaming processes are used on a large scale in the mining industry, for fire fighting and food production, but they can also be limiting factors for improved efficiency in many chemical industries like food and cosmetics production, but also in the oil industry. The experiment FOAM-C (Foam and Optics Mechanics - Coarsening) studies the coarsening process of aqueous wet foam as a function of liquid volume fraction and liquid characteristics. Microgravity allows investigating highly wet foam, which cannot be studied in detail in the earth laboratory due to drainage. Previous experiments have been performed in the frame of the FOAM project, such as parabolic flight campaigns, the Foam-S on the ISS (focused on foams stability) or the experiments in the MAXUS 6 rocket. Now an extensive foam coarsening experiment under microgravity conditions for several types of probes and measurement periods as long as 24 hrs for each sample experiment foam is foreseen to be performed on the ISS.
For the experiment on the ISS, where the storage times before experiment execution can be of several months, one of the crucial development points is the foam generation and quality of the generated foam. The foaming device shall be capable of generating wet foams with different surfactants and with a homogeneous initial size of foam bubbles (<100 µm), as described later in the materials and methods section.
More than 40 samples are foreseen to be processed for the first experiment mission to ISS. A concept of on-orbit replaceable sealed samples has been developed, avoiding the need of a fluid waste reservoir or on orbit sample cleaning.
The foreseen experiment sequence for the on-orbit execution is: 1) ground sample preparation with pre-defined filling procedures, concentrations and liquid fraction, 2) on-orbit foam generation, 3) foam coarsening observation with overview camera, diffusing wave spectroscopy in reflection (DWS) and transmission (DTS) and time resolved correlation spectroscopy (TRC), 4) repeat steps 2 and 3 for all samples is present.
Foam-Coarsening Experiments on the ISS
• 05 August 2020: European science progressed at a slower pace on the International Space Station in the past month. As a series of spacewalks to power up the space habitat came to an end and two of its passengers left for home Earth, intriguing bubbles puzzled researchers and left them wanting to know more. 3)
The Longest Foam
- More bubbles, please. The Space Station has witnessed the longest science run with foams for the Foam-Coarsening experiment. This research mixed varying amounts of water and soap inside small tubes for 14 days in a row. A high-speed piston shook the mixture for two minutes, followed by 100 hours of recording with laser optics, photon counters and high-resolution cameras to achieve the best science.
- The result? The foam bubbles become larger, last longer and their motion is very different from what you would expect on Earth. In space, bubble sizes are evenly spread and that makes it easier for scientists to study them in greater detail.
- In some cases, scientists detected an anomalous behavior in the foam – a hole appeared just in the middle of the bubbles. The team decided to follow up this unexpected phenomenon, and extended the foaming time to up to 20 minutes in some of the tubes.
- The foaming process slows down over time and it is stopped after two weeks, or when there are only five bubbles left inside the tubes. Scientists call it the “five bubble criteria” – this approach helps them estimate when to finish the measurements.
- A foam’s existence in space is marked by more equilibrium than on Earth because drainage is suppressed. Foams have huge benefits for our daily lives – they are lighter, offer better insulation and can be just as strong as compact materials.
- So, maybe think outside your bubble next time you look at a foam, be it in your beer, cream or shaving gel.
- Foam research in microgravity allows researchers to better understand the processes and calculate models in the most optimal conditions. This is leading to better production and assembly of products containing foams as well as more effective foam-suppression agents.
- Many industrial applications benefit from foam research and development in space: cleaning products, cosmetics, fire-fighting and medicines are just some examples. The quality, texture, taste and shelf-life of food and beverages can be enhanced – from the supermarket to the consumers’ fridge.
- "It is a game change for our business" says Cécile Gehin-Delval, from Nestlé Research Laboratories in Orbe, Switzerland.
- The step to space research is closer than you might think. Get involved with spaceflight research via www.esa.int/spaceflightAO. Find out about our commercial partnerships and opportunities in human and robotic exploration via www.esa.int/explorationpartners to run your research in microgravity as well.
• 09 June 2020: The Foam-Coarsening experiment ran a new batch of cartridges in the Fluid Sciences Laboratory of the European Columbus module. 4)
- The experiment began in April to study foams in depth under the more stable conditions afforded by microgravity on the International Space Station.
- The cell cartridges contain a mixture of soap and water. Bubbles are generated by moving a piston at high speed. The foam is observed for up to 100 hours, during which the foam bubbles become larger but less in number. This process slows down over time so measurements are stopped when five bubbles are formed in each cell section. The results are analyzed with laser optics and high-resolution cameras.
- The new batch contains the same water and soap mixture but with a difference in the amount of liquid in the cell. Producing foams in a different concentration allows researchers to study how the bubbles enlarge (or coarsen).
- What is so special about foam? Besides appearing in your food and drink, foams are also found in sealing products, cosmetics and personal hygiene products, and even construction. They are lighter, offer better insulation and can be just as strong as compact materials.
- Observing foams on Earth is tricky because the mixture of gas and liquid that makes up a foam quickly starts to change. Gravity pulls the liquid between the bubbles downwards, and the small bubbles shrink while the larger ones tend to grow at the expense of others. Due to drainage, coarsening (or enlarging) and rupture of the bubbles, a foam starts to collapse back to a liquid state.
- But in space foams are more stable because the liquid does not drain in weightlessness. This allows scientists to study the phenomena of a bubble slowly becoming bigger and bursting, which on Earth are masked by the drainage that destabilizes the foam.
- Deeper insights into the behavior of foams makes for better applications of its use on Earth. Besides improving food production, foams can also be metallic and have incredible structural characteristics. Aluminum foam, for example, is as strong as pure metal but much lighter. This research can help in the construction of light-weight and sturdy aerospace structures and new shielding systems for diagnostic radiology equipment in hospitals.

• 17 March 2020: The Foam-Coarsening experiment, developed by Airbus for ESA, is set to be activated this month but this image (Figure 4) shows that the liquids held in cells are already bubbling as planned. 5)
The image will not be used by the scientists yet but is taken to allow the experiment operators at the Belgian User Operations Centre in Brussels, Belgium, to keep track of the experiment and set it up.
The foams come in self-contained cells and hold liquids that are shaken by pistons and analyzed with laser optics and high-resolution cameras for the scientists on Earth. Researchers are keen to observe how foams behave in microgravity.
On Earth, the mixture of gas and liquid that makes up a foam quickly starts to change. Gravity pulls the liquid between the bubbles downwards, and small bubbles shrink while the larger ones tend to grow at the expense of others. As the liquid is drawn downwards due to gravity the bubbles lose their strength and rupture, collapsing back to a liquid state.
This is annoying for researchers as it limits the time they can study foams and interferes with their experiments. But in space foams are more stable as the liquid does not drain to the bottom in weightlessness.
ESA astronaut Frank De Winne performed the Foam-Stability experiment in 2009 by shaking liquid solutions and recording what happened next. The samples ranged from pure water to protein-based fluids, like the ones used for chocolate foams, and antifoaming agents.
After just ten seconds, the fluids stabilized more quickly and produced more foam than on Earth. Scientists discovered that it was possible to create super-stable foams in zero gravity.
Building on this extensive foam research, Foam-Coarsening will investigate foam behavior at different liquid stages, particularly as it transitions from a solid-to liquid-like state.
The results from this research will not just apply to the foam in your morning cappuccino. Foams are used in a wide range of areas from food production to cleaning and sealing products, cosmetics and personal hygiene products, and even construction.
NASA astronaut Jessica Meir installed the experiment in the Fluid Science Laboratory on 6 March after removing the Multiscale boiling experiment known as Rubi. The experiment is controlled and data collected by the Belgian User Operations Centre (BUOC) who processed this image on 9 March.

Aqueous Foams and Foam Films Stabilized by Surfactants. Gravity-free Studies
Foams are dispersions of gas bubbles in liquids or solid matrices. They occur widely in nature and are used in important industrial processes and commercial applications. Liquid foams are an essential part of gas/liquid contacting processes such as distillation and flotation of minerals. Foams made with aqueous surfactant solutions are widely used for detergency purposes, while foams made with protein solutions are the base of many food products. Other applications of liquid foams include cosmetics, fire-fighting, oil recovery, and others. Solid polymeric foams, such as polystyrene and polyurethane, find applications as insulation panels in the construction industry and as packing and cushioning materials. Solid foams can also be made of metals, nowadays used in car and space industries 6)
Foams are usually prepared with liquid matrices. Because of the large density difference between air and the liquid, this liquid drains rapidly due to gravity and dry foams are obtained. Great progress has been made over the last thirty years in making the science of dry foams a sound and systematic discipline, with theories based on well-defined models being compared with well-characterized experiments. Properties of interest have included: the details of foam structure(shapes and arrangements of bubbles), capillary and gravity driven drainage, rheology, coarsening due to diffusion of gas (growth of large bubbles, shrinkage of small ones) and coalescence due to rupture of the films between bubbles.
Foam dynamics involves a variety of phenomena occurring at different length scales: molecular (monolayers of surface active agents at the air–water interface, thickness ~1–10 nm), mesoscopic (films between bubbles, thickness ~10 nm–1 µm), and macroscopic (bubbles and channels between bubbles called Plateau borders, ~100 µm – 1 cm). These phenomena occur at different timescales, typically microseconds to hours. Furthermore, coarsening and coalescence are coupled with gravity drainage and are faster when the foam liquid fraction is lower. Due to this complexity, we still know little about coarsening and coalescence, a serious drawback for the prediction of dry foam stability. The study of the behavior of the foam in the early stages, when it still contains large amounts of liquid, remains still more elusive. As a consequence, it is difficult to predict what will be the final foam structure although this knowledge would help to better target the applications.
All these open questions have motivated foam investigations in microgravity. Microgravity is indeed a helpful tool to suppress drainage and gain more insight into the coarsening and coalescence mechanisms and into the behavior of wet foams. In this paper, we will briefly summarize the work that we have done over the years thank to the support of CNES, in order to elucidate aqueous foams and foam films behavior. We will focus on aqueous foams stabilized by surfactants. Part of this work has also been supported by ESA through a MAP program. Various experiments were performed in gravity-free conditions; others are scheduled in the near future. In the following, we will first recall some general concepts in foam science, then describe the different experiments and end with the plans for the near future.
General Concepts for Foams
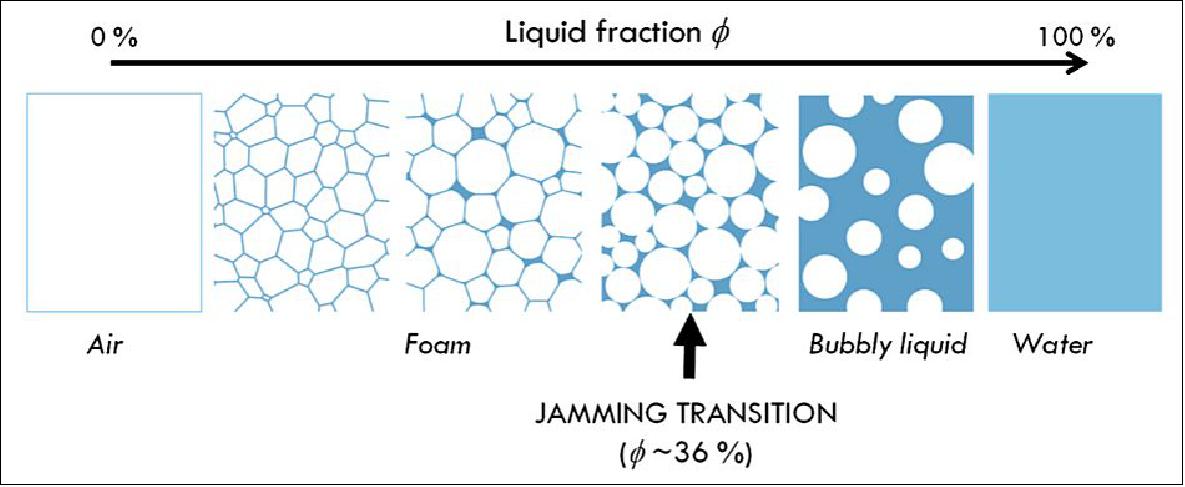
The liquid volume fraction in foam, φ, may vary from less than one percent (dry foam) to more than 10% (wet foam) (Figure 5). At low liquid fractions, the bubbles are deformed into polyhedra with both quasi-flat faces (bordering films) and curved faces (at the edges). When φ is larger than a critical value φc(~35% for disordered foams and 25% for ordered ones), the bubbles are spherical and move independently; in this case, the term bubbly liquid is used instead of foam. An interesting transition occurs at φc where the foam changes from solid-like (finite shear modulus G) to liquid-like (disconnected bubbles). This is the jamming transition, also encountered in other assemblies of randomly packed objects, such as emulsions, sand, clays, etc., and presently the object of numerous studies. 7)
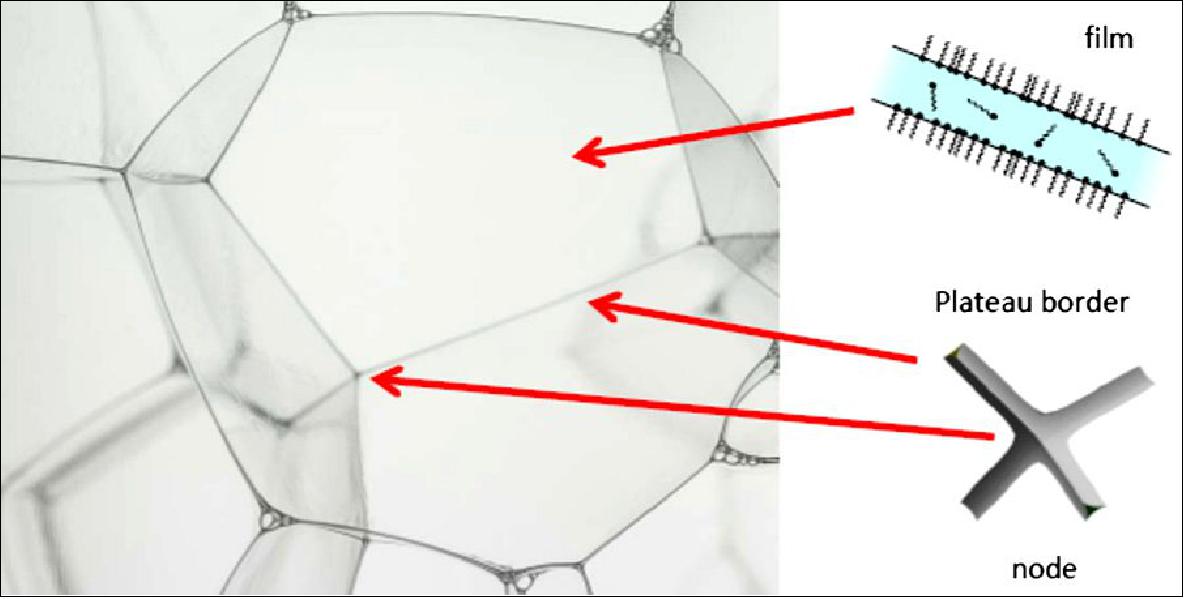
In order to generate a foam, an energy is needed to create the bubble surfaces, equal to γ A, γ being the surface tension and A the area created. This energy is orders of magnitude larger than thermal energies and is not minimized: as a consequence, liquid foams are thermodynamically unstable. However, metastable configurations can be produced, in which each bubble takes a shape having minimal area for the given configuration: spheres for isolated bubbles and polyhedra (like the well-known tetrakaidecahedron proposed by Kelvin) in foams with small liquid volume fractions. Most of the liquid is confined to the edges called Plateau borders(PB), connecting three films and junctions or nodes connecting four PBs (Figure 6).
Because liquid foams are metastable, the use of stabilizing agents is required, which are generally surfactant molecules (Figure 6), but polymers, proteins or particles can also be used. The stabilizer role is to slow down the different mechanisms of foam ageing: drainage, coalescence and coarsening. After formation, foams evolve with time until they fully disappear, typically a few hours later with surfactant foams. The foam destruction starts with the rupture of foam films, hence foam films have also been extensively studied in link with foam stability.
Experiments on Foam Films
We started our studies with model foam films, an activity supported by CNES in the 1990s. It was easy in this case to simulate microgravity conditions by studying horizontal films. We constructed devices similar to those proposed by Sheludko [8)] and Mysels [9)], later called thin film balances(TFBs). In the Sheludko device, horizontal films are formed in capillaries connected to a liquid reservoir of controlled hydrostatic pressure. The film thickness is measured by optical reflectivity. The Mysels’ device is similar, but it makes use of a porous plate instead of a capillary tube, allowing accessing much larger pressures. In both cases, the pressure in the film holder is lower than the pressure in the film center by a quantity ΔP. As a result and despite the absence of gravity force, the films drain because of the imposed pressure difference ΔP. Note that horizontal foam films also drain, because in this case, the pressure difference is the capillary pressure arising from the PB’s curvature (Figure 6). This type of drainage is called capillary drainage.
Experiments on Foams
Foam drainage: Gravity plays an important role in the formation of foam and its subsequent evolution. Its primary effect is to cause excess liquid to drain rapidly away. When the foam is stable enough, it becomes dry and the gravitational force is balanced by a vertical pressure gradient in the liquid leading to a vertical profile of liquid fraction. 10) The liquid volume fraction φ at the top of the foam are typically smaller than a few percent.
The addition of liquid at the top of such a dry foam results in a solitary wave with an approximately constant profile during its downward passage, the process being called forced drainage. When we started our first foam studies, there were two theories of forced drainage, one assuming rigid surfaces and the other mobile interfaces, without obvious reason allowing deciding which one was correct.
Surface foams: The stability of aqueous foams made with various surfactants both on Earth and in the ISS.

Generally, the foamability was found much larger on the ISS than on Earth. A striking result is that solutions that foam little on Earth lead to significant amounts of foam in space. Moreover, the foams showed a remarkable stability in the ISS. The foams were quite wet, with liquid fractions φ of 30%. The bubbles had a quasi-spherical shape which slows down capillary drainage, coarsening and coalescence.
References
1) ”Foam-Coarsening experiment,” ESA Science & Exploration, 9 March 2020, URL: http://www.esa.int/ESA_Multimedia/Images/2020/03/Foam-Coarsening_experiment
2) Rodrigo Carpy, Gerold Picker, Benjamin Amann, Hans Ranebo, Sébastien Vincent-Bonnieu, Olivier Minster, Josef Winter, Jan Dettmann, Luigi Castiglione, Reinhard Höhler, Dominique Langevind, ”Foam Generation and Sample Composition Optimization for the FOAM-C Experiment of the ISS,” Journal of Physics: Conference Series Volumr 327 (2011) 012025, URL: https://iopscience.iop.org/article/10.1088/1742-6596/327/1/012025/pdf
3) ”Weightless action on the Space Station – power, bones and bubbles,” ESA Science & Exploration, 05 August 2020, URL: https://www.esa.int/Science_Exploration/Human_and_Robotic_Exploration
/Weightless_action_on_the_Space_Station_power_bones_and_bubbles
4) ”Foam, of coarse,” ESA Science & Exploration, 09 June 2020, URL: http://www.esa.int/ESA_Multimedia/Images/2020/06/Foam_of_coarse
5) ”Space foam,” ESA Science & Exploration, 17 March 2020, URL: http://www.esa.int/ESA_Multimedia/Images/2020/03/Space_foam
6) Dominique Langevin, ”Aqueous foams and foam films stabilized by surfactants. Gravity-free studies,” Comptes Rendus Mécanique, Volume 345, Issue 1, January 2017, Pages 47-55, https://doi.org/10.1016/j.crme.2016.10.009, URL: https://tinyurl.com/srz4rrr
7) Andrea J. Liu and Sidney R. Nagel, ”The Jamming Transition and the Marginally Jammed Solid,” Annual Review of Condensed Matter Physics,Vol. 1:347-369 (Volume publication date 2010), First published online as a Review in Advance on May 20, 2010, https://doi.org/10.1146/annurev-conmatphys-070909-104045
8) A. Sheludko, ”Thin liquid films,” Advances in Colloid and Interface Science, Volume 1, Issue 4, December 1967, Pages 391-464, Available online 7 August 2001, https://doi.org/10.1016/0001-8686(67)85001-2
9) Karol J. Mysels and Malcolm N. Jones,”Direct Measurement of the variation of double-layer repulsion with distance,” Discussions of the Faraday Society, Vol. 42, 1966, pp: 42-50, DOI: 10.1039/DF9664200042
10) Armando Maestro, Wiebke Drenckhan, Emmanuelle Rioa and Reinhard Höhler, ”Liquid dispersions under gravity: volume fraction profile and osmotic pressure,” Soft Matter, Vol. 9, Issue 8, 2013, pp. 2531-2540, DOI: 10.1039/c2sm27668b
The information compiled and edited in this article was provided by Herbert J. Kramer from his documentation of: ”Observation of the Earth and Its Environment: Survey of Missions and Sensors” (Springer Verlag) as well as many other sources after the publication of the 4th edition in 2002. - Comments and corrections to this article are always welcome for further updates (eoportal@symbios.space).